|
They are formed in a multidimensional network of solution equilibria involving electron transfer and contact ion pair formation as well as aggregation, and are often dominated by cation solvation such solvation is known to control numerous chemical reactions, including geochemical and biochemical reactions. Molecular anions M n - and their countercations Met solv (where Met is the alkali metal) crystallize from aprotic solvents either as solvent-separated n or as solvent-shared solv ion pairs. 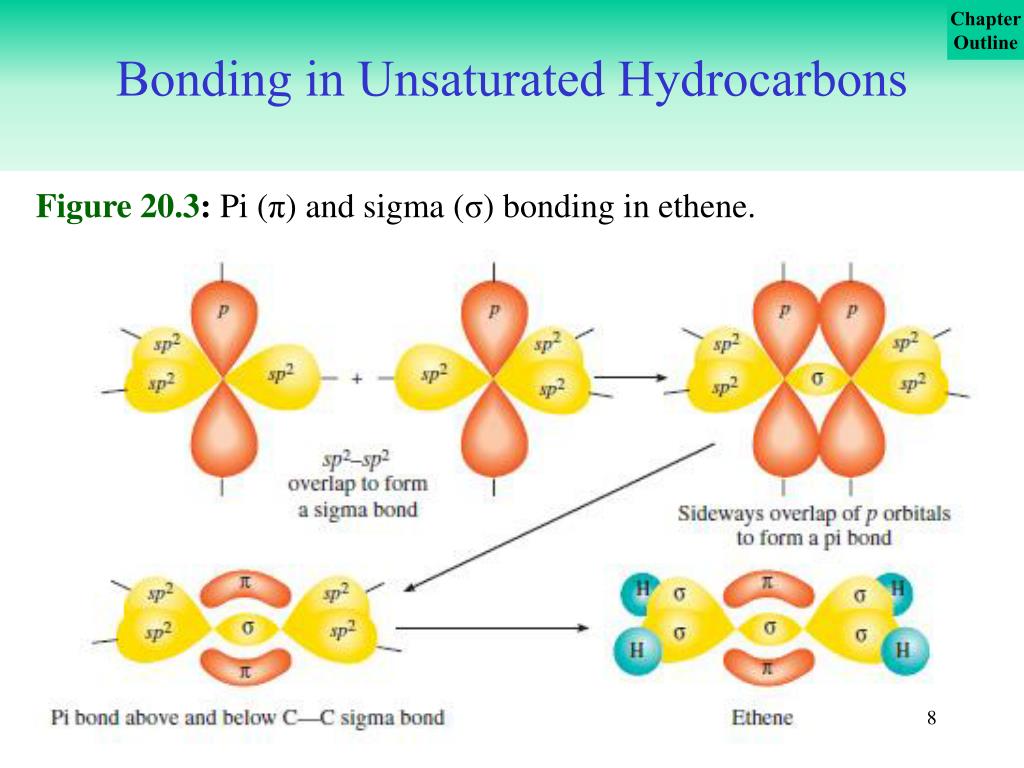
We expect that the successful crystallization of the ionic solid we report here, and that of a covalent organic compound with a triplet ground state at room temperature, will stimulate further attempts to develop new triplet-ground-state materials for practical use. Although the spins in this biradical ionic solid are separated by a considerable distance, density functional theory calculations indicate that the triplet ground state is 84 kJ mol -1 more stable than the first excited singlet state. Its electronic ground state is shown experimentally, using temperature-dependent electron paramagnetic resonance spectroscopy, to be a triplet. 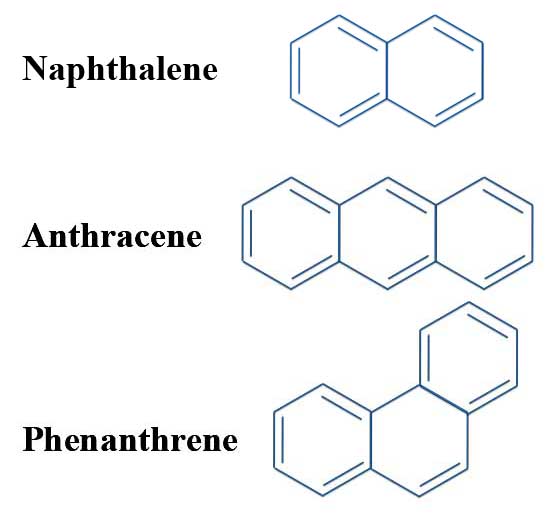
Here we report the anaerobic crystallization of an ionic organic aggregate-a contact ion septuple consisting of a fourfold negatively charged 'tripledecker' of three anthracene molecules bridged by four solvated potassium cations. The formation of supramolecular aggregates can even be exploited to generate aromatic hydrocarbons that carry four negative charges and crystallize in the form of organic poly(metal cation) clusters or helical polymers. Aggregates containing metal cation clusters 'wrapped' by lipophilic molecular anions have, for example, been shown to be kinetically stable and soluble in nonpolar liquids such as saturated hydrocarbons. Havlas Ĭrystalline supramolecular aggregates consisting of charged organic molecules, held together through metal-cluster-mediated Coulomb interactions, have attracted interest owing to their unusual structural, chemical and electronic properties. For this reason, the Hückel method is limited to planar systems.Author(s): H. This is referred to as sigma-pi separability and is justified by the orthogonality of \(\sigma\) and \(\pi\) orbitals in planar molecules. 
The method limits itself to addressing conjugated hydrocarbons and specifically only \(\pi\) electron molecular orbitals are included because these determine the general properties of these molecules the sigma electrons are ignored. The wavefunctions used to describe the bonding orbitals in each framework results from different combinations of atomic orbitals. Within the Hückel approximation, the covalent bonding in these hydrocarbones can be separated into two independent "frameworks": the \(\sigma\)-bonding framework and the the \(\sigma\)-bonding framework. The Hückel approximation is used to determine the energies and shapes of the \(\pi\) molecular orbitals in conjugated systems. (Note: by convention, in planar molecules the axis perpendicular to the molecular plane is the z-axis.) (CC BY-NC Ümit Kaya via LibreTexts) (b) One singly occupied unhybridized 2p z orbital remains on each carbon atom to form a carbon–carbon \(π\) bond. This uses 10 of the 12 valence electrons to form a total of five \(σ\) bonds (four C–H bonds and one C–C bond). : (a) The σ-bonded framework is formed by the overlap of two sets of singly occupied carbon sp 2 hybrid orbitals and four singly occupied hydrogen 1s orbitals to form electron-pair bonds. Thus each carbon forms a set of three \(\sigma\) bonds: two C–H ( sp 2 s) and one C–C ( sp 2 sp 2) (part (a) of Figure 10.5.1 This angle suggests that the carbon atoms are sp 2 hybridized, which means that a singly occupied sp 2 orbital on one carbon overlaps with a singly occupied s orbital on each H and a singly occupied sp 2 lobe on the other C. Experimentally, we know that the H–C–H and H–C–C angles in ethylene are approximately 120°. The simplest hydrocarbon to consider that exhibits \(\pi\) bonding is ethylene (ethene), which is made up of four hydrogen atoms and two carbon atoms. This is, in fact, a more sophisticated version of a free-electron model. An approximation introduced by Hückel in 1931 considers only the delocalized p electrons moving in a framework of \(\pi\)-bonds. Molecular orbital theory has been very successfully applied to large conjugated systems, especially those containing chains of carbon atoms with alternating single and double bonds. Demonstrate how Hückel's theory approximates the full molecular orbital picture of molecules by treating the \(\sigma\)-bonding and \(\pi\)-bonding networks independently.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
